
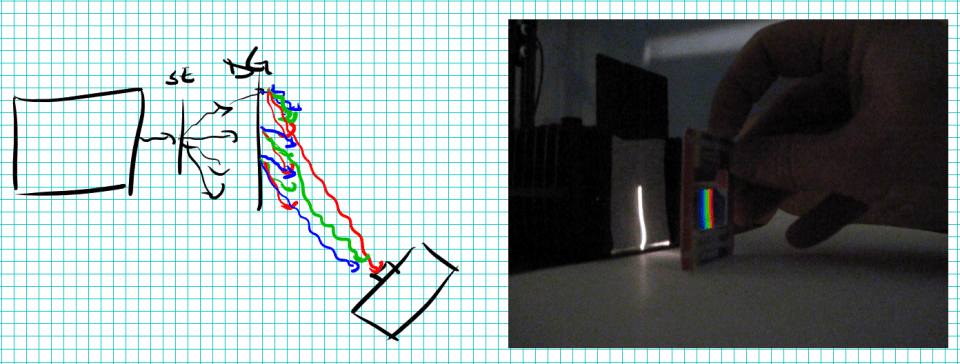
Fluorescence is amazing. You can take a cheap dye, attach it to almost anything, and then detect that thing at absurdly low concentrations. Fluorescein is the dye in the picture above. I hit it with my blue LED illuminator, and it glows with an intense green light. I suspect that basically every glowing green sciencey thing in every movie ever was really just fluorescein. It’s also the dye that they used to use to turn the boston river green. It’s also injected into human veins for an angiogram. Back in the day, I used a powerful laser confocal microscope to see individual fluorescein molecules.
Anyway, I love fluorescein. I’m going to use it today to talk about detection limits.
The other day I was talking about how “the dose makes the poison.” Some folks get all up in arms because of small amounts of (for example) formaldehyde. Chronic exposure to high levels of formaldehyde is bad. Acute exposure to gram-quantities could kill. But a single exposure to a microscopic quantity is not dangerous. It’s highly reactive and will be destroyed by formaldehyde dehydrogenase very rapidly.
When I talked about any level of formaldehyde being safe (it’s a metabolic byproduct! Your body knows how to deal!) I got accused of being a shill for “Big Pharma” (sigh). I tell you what: I wish it was that easy to get money from a pharmaceutical company. I say simple factual information, and they cut a check? Yes please!
I digress. The point is that just because we can’t detect it doesn’t mean it’s not there. And just because we can detect it doesn’t mean it’s dangerous. We get better at detecting things every year. That doesn’t mean the world is getting more hostile. We’re going to talk about quantifying the limits of our measurements today in my instrumental analysis class.
I put up a video honoring Friday the 13t superstitions. It has a movie of that fluroescein drop at the end and I’m pretty impressed with myself for how cool it looks.




You must be logged in to post a comment.